Using Human-Induced Pluripotent Stem Cells to Investigate a Novel CPVT-Related RyR2 Variant
Main Article Content
Abstract
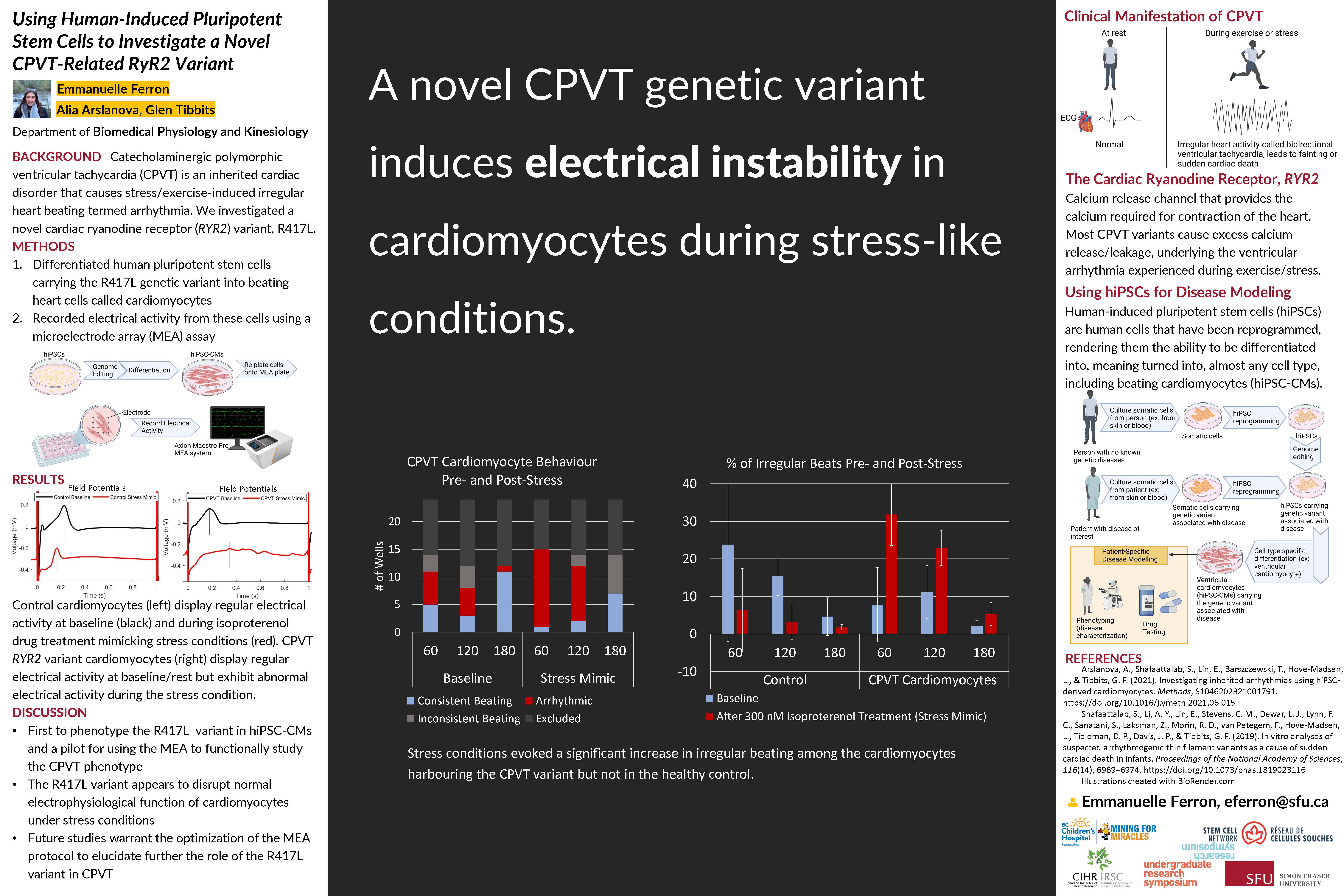
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited cardiac disorder that results in stress/exercise-induced irregular heart beating known as ventricular arrhythmia. Most CPVT cases have been linked to genetic variants in the cardiac ryanodine receptor (RYR2). We introduced a novel RYR2 genetic variant, R417L, that has been associated with CPVT into human-induced pluripotent stem cells (hiPSCs). HiPSCs can be turned into a multitude of different cell types through a process called differentiation. We differentiated these hiPSCs into beating ventricular cardiomyocytes which are muscular heart cells. To study the electrical properties of these cells, we used a microelectrode array assay (MEA) which uses a grid of electrodes to detect and record the electrical activity of the overlying cells during beating. The MEA can also stimulate the cells to train them to beat at a certain frequency. The goal of our research was to recapitulate the CPVT phenotype in our differentiated ventricular cardiomyocytes carrying the CPVT genetic variant and optimize the MEA protocol for research and drug testing on CPVT pathogenic variants. Initial experimentation found that stress/exercise conditions resulted in a significant increase in the number of arrhythmic wells among the CPVT variant cardiomyocytes. However, these results were not replicated in two subsequent trials using the same cells. Proper electrode coverage, stimulation parameters and health of the cells remain to be optimized for future research using the MEA to investigate CPVT variants. Gaining insight into this novel CPVT variant may have important implications towards improving the treatment of patients with CPVT.
Faculty Supervisor:
Dr. Glen Tibbits, Biomedical Physiology and Kinesiology, Simon Fraser University
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.